SolutionHard
Question
The molecular weight of benzoic acid in benzene as determined by determined by depression in freezing point method corresponds to :
Options
A.ionisation of benzoic acid
B.dimerisation of benzoic acid
C.trimerisaton of benzoic acid
D.solvation of benzoic acid
Solution
Due to intermolecular H-bonding benzoic acid remains in dimeric state.
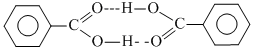
Create a free account to view solution
View Solution FreeMore Solution Questions
The freezing point among the following equimolal aqueous solutions, will be highest for :...Thevan′t Hoff factor, i for a compound which undergoes dissociation in one solvent and association in other solven...A complex containing K+, Pt(IV) and Cl- is 100% ionised giving i = 3. Thus, complex is :...At 35°C, the vapour pressure of CS2 is 512 mm Hg and that of acetone CH3COCH3 is 344 mm Hg. A solution of CS2 and aceton...Which one is not equal to zero for an ideal solution:-...