Atomic StructureHard
Question
The electron in a hydrogen atoms makes a transition n1 → n2, n1 and n2 are the principal quantum number of two states. Assume the Bohr′s model to be valid. The time period of the electrons in the initial state is eight times that in the final state. The possible values of n1 and n2 are
Options
A.n1 = 4, n2 = 2
B.n1 = 8, n2 = 2
C.n1 = 8, n2 = 1
D.n1 = 6, n2 = 3
Solution
Time period, Tn = 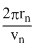
i.e., Tn ∝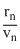
But rn ∝ n2 and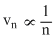
Therefore, Tn ∝ n3
Given, Tn1 = 8Tn2
Hence, n1 = 2n2
Therefore, options (a) and (d) are correct.
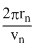
i.e., Tn ∝
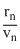
But rn ∝ n2 and
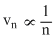
Therefore, Tn ∝ n3
Given, Tn1 = 8Tn2
Hence, n1 = 2n2
Therefore, options (a) and (d) are correct.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The fossil bone has a 14C : 12C ratio, which is f that in a living animal bone. If the half life time of 14C is 5730 yea...The plate resistance of a triod is 3 × 103Ω and its mutual conductance is 1.5 × 10-3 A/V. The amplificati...A shell is fired from a cannon with velocity n m/sec at an angle θ with the horizontal direction. At the highest po...An electrons of a stationary hydrogen atom passes from the fifth energy level to the ground level. Thevelocity that the ...During a negative beta decay...