Atomic StructureHard
Question
A stationary hydrogen atom of mass M emits a photon corresponding to the first line of lyman series. If R is the Rydberg′s constant, the velocity that the atom acquires is :-
Options
A.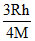
B.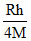
C.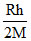
D.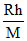
Solution
Momentum of H atom = Momentum of photon.
mv =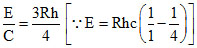
v =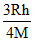
mv =
v =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In the Bohr model of the hydrogen atom...If a source of power 4 kW produces 1020 photons/second, the radiation belong to a part of the spectrum called...Hydrogen (1H1), Deuterium (1H2) singly ionised Helium (2He4)+ and doubly ionised lithium (3Li6)++; all have one electron...A radiation of energy E falls normally on a perfectly reflecting surface. The momentum transferred to the surface is...freshly prepared radioactive source of half-life 2 h emits radiation of intensity which is 64 times of the permissible s...