Atomic StructureHard
Question
In the Bohr model of the hydrogen atom
Options
A.the radius of the nth orbit is proportional n2
B.the total energy of the electron in the nth orbit is inversely proportional to n
C.the angular momentum of the electron in an orbit is an integral multiple of h/π
D.the magnitude of the potential energy of the electron in any orbit is greater than its kinetic energy
More Atomic Structure Questions
The potential difference applied to an X-ray tube is 5 kV and the current through it is 3.2 mA. The number of electrons ...The surface of zone material is radiated in turn by waves of 10λ = 350nmand 540 nm respectively. The ratio of the s...As per Bohr's model, the minimum energy (in eV) required to remove an electron from the ground state of doubly ionized L...The energy of a photon of light with wavelength 5000 is approximately 2.5 eV. This way the energy of an X-ray photon wit...Beta rays emitted by a radioactive material are...
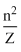 and |PE| = 2(KE)
and |PE| = 2(KE)