Nuclear Physics and RadioactivityHardBloom L3
Question
The half-life of $^{215}At$ is $100\ \mu s$. The time taken for the activity of a sample of $^{215}At$ to decay to $\dfrac{1}{16}$th of its initial value is: 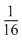
Options
A.$400\ \mu s$
B.$63\ \mu s$
C.$40\ \mu s$
D.$300\ \mu s$
Solution
{"given":"Half-life $T_{1/2} = 100\\ \\mu s$, final activity $R = \\dfrac{R_0}{16}$","key_observation":"Activity decays as $R = R_0 \\left(\\dfrac{1}{2}\\right)^n$ where $n = \\dfrac{t}{T_{1/2}}$ is the number of half-lives elapsed.","option_analysis":[{"label":"(A)","text":"$400\\ \\mu s$","verdict":"correct","explanation":"Setting $\\left(\\frac{1}{2}\\right)^n = \\frac{1}{16} = \\left(\\frac{1}{2}\\right)^4$ gives $n = 4$, so $t = 4 \\times 100\\ \\mu s = 400\\ \\mu s$."},{"label":"(B)","text":"$63\\ \\mu s$","verdict":"incorrect","explanation":"This value does not correspond to any integer number of half-lives and does not satisfy the decay equation for $R = R_0/16$."},{"label":"(C)","text":"$40\\ \\mu s$","verdict":"incorrect","explanation":"This corresponds to $n = 0.4$ half-lives, giving $R/R_0 \\approx 0.76$, not $1/16$."},{"label":"(D)","text":"$300\\ \\mu s$","verdict":"incorrect","explanation":"This corresponds to $n = 3$ half-lives, giving $R = R_0/8$, not $R_0/16$."}],"answer":"(A)","formula_steps":[]}
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
How many head - on elastic collisions must a neutron have with deuterium nuclei to reduce it energy from 6.561 MeV to 1 ...A radio isotope X with a half life 1.4 × 109 years decays of Y which is stable. A sample of the rock from a cave wa...How much energy will be released when 10 kg of U235 is completely converts into energy :-...Decay rate is proportional to :-...The above is a plot of binding energy per nucleon Eb, against the nuclear mass M; A, B, C, D, E, F correspond to differe...