Ionic EquilibriumHard
Question
Let the colour of the indicator HIn (colourless) will be visible only when its ionised form (pink) is 25% or more in a solution. Suppose HIn (pKa = 9.0) is added to a solution of pH = 9.6 predict what will happen. (Take log 2 = 0.3)
Options
A.pink colour will be visible
B.pink colour will not be visible
C.% of ionised form will be less than 25%
D.% of ionised form will be more than 25%
Solution
HIn → H+ + In-
KIn =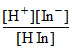 ⇒
⇒ 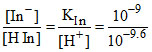 = 100.6 = 4
= 100.6 = 4
∴ % of [In-] in solution =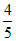 × 100 = 80%
× 100 = 80%
so pink colour will be visible.
KIn =
∴ % of [In-] in solution =
so pink colour will be visible.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of a 0.1 M monobasic acid is found to be 2. Hence its osmotic pressure at a given temperature TK is :-...Which one of the following statements is not true?...When excess oxalic acid is added to CaCl2 solution, CaC2O4 is precipitated and the solution still contains some unprecip...The best explanation for the solubility of MnS in dilute HCl is that :...Which of the following solution will have a pH exactly equal to 8 ?...