Atomic StructureHard
Question
The respective values of orbital angular momentum of an electron in 1s, 2s, 3d and 3p-orbitals are:-
Options
A.0, 0, √6 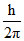 , √2
, √2 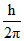
B.1, 1, √4 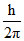 , √2
, √2 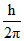
C.0, 1, √6 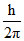 , √3
, √3 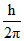
D.0, 0, 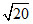
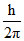 , √6
, √6 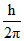
More Atomic Structure Questions
When lithium vapours were filled in the discharge tube for anode rays experiment, the anode rays were found to contain o...A photon of 2.55 eV is emitted out by an electronic transition in hydrogen atom. The change in de Broglie wavelength of ...The ratio of energies of first excited state of He+ ion and ground state of H–atom is...Photon having energy equivalent to the binding energy of 4th state of He+ ion is used to eject an electron from the meta...In an experiment, 4g of M2Ox oxide was reduced to 2.8 g of the metal. If the atomic mass of the metal is 56 g mol-1. The...