Atomic StructureHard
Question
Which of the following compounds, conjugate base is stablized by d-orbital resonance -
Options
A.Cl3C − CH2CO2H
B.Cl3C − CO2H
C.Cl3CH
D.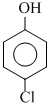

Solution
HCCl3 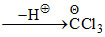
Here Θ charge is stabilized by d-orbital resonance as Cl has vacant d-orbital.
Here Θ charge is stabilized by d-orbital resonance as Cl has vacant d-orbital.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A rod PQ of mass M and length L is hinged at end P. The rod is kept horizontal by a massless string tied to point Q as s...A sodium street light gives off yellow light that has a wavelength of 600 nm. Then (For energy of a photon take E = )...The wavelength of the radiation emitted, when in hydrogen atom electron falls from infinity to stationary state 1, would...If wavelength is equal to the distance travelled by the electron in one second, then -...The element of the Lanthenoide series have maximum number of unpaired electron :-...