Atomic StructureHard
Question
The wavelength of the radiation emitted, when in hydrogen atom electron falls from infinity to stationary state 1, would be (Rydberg constant = 1.097 × 107 m-1)
Options
A.91 nm
B.9.1 × 10-8 nm
C.406 nm
D.192 nm
More Atomic Structure Questions
The frequency of radiation emitted when the electron falls from n = 4 to n = 1 in a hydrogen atom will be(Given h = 6.62...How many of the following compounds have used their d-orbital in hybridisationCF4, SF4, I3-, SOF4, PF3Cl2, SnCl2, SO42-...Which of the following pairs of compounds is isoelectronic and isostructural ?...Biotite has the emperical formula of (Si2O5)x2x- . Find the number of oxygen atom shared per tetrahedron...A proton and an a-particle are accelerated through the same potential difference from rest. Then the ratio of their de B...
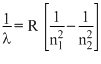
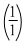
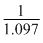 × 10-7 m
× 10-7 m