Gaseous StateHard
Question
Consider the equation Z = 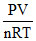 which of the following statement is correct ?
which of the following statement is correct ?
Options
A.When Z > 1 real gases are easier to compress than the ideal gas
B.When Z = 1 real gases get compressed easily
C.When Z > 1 real gases are difficult to compress
D.None
More Gaseous State Questions
At high pressure, the compressibility factor (Z) is equal to :-...The rate of diffusion of methane at a given temperature is twice that of a gas X. The molecular weight of X is :...A certain volume of argon gas (Mol. Wt. = 40) requires 45 s to effuse through a hole at a certain pressure and temperatu...Consider given reaction : Metal (M) Select correct statement(s)...According to the kinetic theory of gases, in an ideal gas, between two successive collisions a gas molecule travels...