Gaseous StateHard
Question
The rate of diffusion of methane at a given temperature is twice that of a gas X. The molecular weight of X is :
Options
A.64.0
B.32.0
C.4.0
D.8.0
Solution
According to Graham,s law, 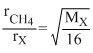
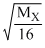 = 2 ⇒ MX = 64
= 2 ⇒ MX = 64
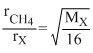
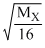 = 2 ⇒ MX = 64
= 2 ⇒ MX = 64Create a free account to view solution
View Solution FreeMore Gaseous State Questions
1g H2 gas at STP is expanded so that volume is doubled. Hence work done is :-...At what temperature, the average speed of gas molecules be double of that at temperature, 27oC?...An amount of 1.00 g of a gaseous compound of boron and hydrogen occupies 0.820 liter at 1.00 atm and at 3oC. The compoun...A mixture of hydrogen and oxygen at one bar pressure contains 20% by weight of hydrogen. Partial pressure of hydrogen wi...An element (atomic mass 100g/mol) having BCC structure has unit cell edge 400 pm. the density of element is (No. of atom...