Gaseous StateHard
Question
At high pressure, the compressibility factor (Z) is equal to :-
Options
A.1
B.1 - 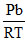
C.1 + 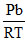
D.Zero
Solution
At high pressure.
P(V - b) = RT
PV - Pb = RT
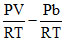 = 1
= 1
Z = 1 +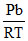
P(V - b) = RT
PV - Pb = RT
Z = 1 +
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
If the pressure of a gas contained in a closed vessel is increased by 0.4 % when heated by 1oC its initial temperature m...An amount of 1.00 g of a gaseous compound of boron and hydrogen occupies 0.820 liter at 1.00 atm and at 3oC. The compoun...The ratio between the r.m.s. velocity of H2 at 50 K and that of O2 at 800 K is:...At constant pressure which of the following does not represent Charle′s law ?...Amongst the following statement, the correct one is:...