Atomic StructureHard
Question
The speed of an electron having a wavelength of 10-10 m is
Options
A.4.24 × 106 m/s
B.5.25 × 106 m/s
C.6.25 × 106 m/s
D.7.25 × 106 m/s
Solution
We know that De-Broglie wavelength is
mv =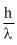 ⇒ λ =
⇒ λ = 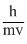
10-10 =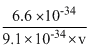
v =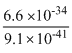 = 7.25 × 106
= 7.25 × 106
mv =
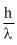 ⇒ λ =
⇒ λ = 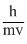
10-10 =
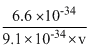
v =
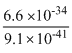 = 7.25 × 106
= 7.25 × 106 Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following statement(s) is (are) correct ?...If a physical quantity A associated with a hydrogen atom satisfies: = constant, where r is the radius of the electronic ...The operation of a nuclear reactor is said to be critical, if multiplication factor (K)has a value...If the binding energy of the electron in a hydrogen atom is 13.6 eV, the energy required to remove the electron from the...Solid targets of different elements are bombarded by highly energetic electron beam. the frequency (f) of the characteri...