ThermodynamicsHard
Question
An ideal gas is taken through a process where dQ = dU - dW. The specific heat of the ideal gas in this process is (dQ = heat supplied to the gas, dU = change in internal energy, dW = work done by gas)
Options
A.Cv
B.Cv + 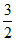 R
R
C.Cv - R
D.the process is impossible
Solution
dQ = dU - dW, again dQ = dU + dW
⇒ dW = pdV = 0
⇒ dW = pdV = 0
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The average translational kinetic energy of O2 (molar mass 32) molecules at a particular temperature is 0.048 eV. The tr...2 gm of O2 gas is taken at 27oC and pressure 76cm. of Hg. Then find out volume of gas - (in litre)...In a water fall the water falls from a height of 100 m. If the entire energy of water is converted into heat. The rise i...A gas for which γ = 5/3 is heated at constant pressure. The percentage of total heat given that will be used for ex...A carnot′′s engine operates between 400 K and 800 K, its efficienty is...