Chemical Kinetics and Nuclear ChemistryHard
Question
Arrange following compounds in decreasing order of reactivity for hydrolysis reaction :-
(I) C6H5COCl
(II) NO2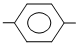 COCl
COCl
(III) CH3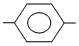 COCl
COCl
(IV) OHC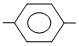 COCl
COCl
(I) C6H5COCl
(II) NO2
 COCl
COCl(III) CH3
 COCl
COCl(IV) OHC
 COCl
COClOptions
A.II > IV > I > III
B.II > IV > III > I
C.I > II > III > IV
D.IV > III > II > I
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate constant, the activation energy and the Arrhenious parameter of a chemical reaction at 25oC are 3.0 × 10-4...A sample of 53I131, as I- ion, was administered to a patient in a carrier consisting 1.0 mg of stable I- ion. After 4.0 ...Which of the following nuclear reactions will generate an isotope?...A substance ″A″ decomposes in solution following the first order kinetics. Flask I contains 1 L of 1 M solut...The rate of reaction A + B → Products is given by the equation r = k[A][B]. If B is taken in large excess, the ord...