ThermodynamicsHard
Question
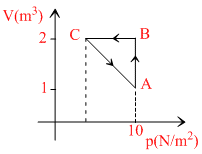
An ideal gas is taken through the cycle A → B → C → A, as shown in the figure. If the net heat supplied to the gas in the cycle is 5 J, the work done by the gas in the process C → A is

Options
A.- 5 J
B.- 10 J
C.- 15 J
D.- 20 J
Solution
ᐃWAB = pᐃV = (10) (2 - 1) = 10 J
WBC = 0 (as V = constant)
From first law of thermodynamics
dQ = dU + dW
dU = 0 (process ABCA is cyclic)
∴ ᐃQ = ᐃWAB + ᐃWBC + ᐃWCA
∴ ᐃWCA = ᐃQ - ᐃWAB - ᐃWBC
= 5 - 10 - 0 = - 5J.
WBC = 0 (as V = constant)
From first law of thermodynamics
dQ = dU + dW
dU = 0 (process ABCA is cyclic)
∴ ᐃQ = ᐃWAB + ᐃWBC + ᐃWCA
∴ ᐃWCA = ᐃQ - ᐃWAB - ᐃWBC
= 5 - 10 - 0 = - 5J.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
If the heat of 110 J is added to a gaseous system, change in internal energy is 40 J, then the amount of external work d...A black body is at temperature if 2800 K. The energy of radiation emitted by this body with wavelength between 499 nm an...During isothermal, isobaric and adiabatic processes, work done for same change in volume will be maximum for :-...During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its absolute temperature. ...The temperature of an ideal gas is increased from 120 K to 480 K. If at 120 K theroot mean square velocity of the gas mo...