ThermodynamicsHard
Question
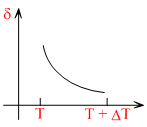
An ideal gas is initially at temperature T and volume V. Its volume is increased by ᐃV due to an increase in temperature ᐃT, pressure remaing constant. The quantity δ = ᐃV /VᐃT varies with temperature as
Options
A.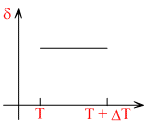

B.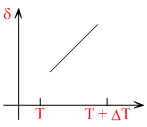

C.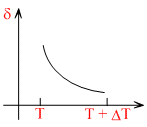

D.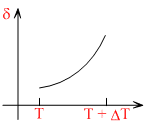

Solution
For an ideal gas : pV = nRT
For p = constant
pᐃV = nRTᐃT
∴
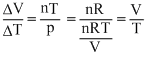
∴
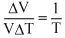 or δ =
or δ = 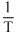
Therefore, δ is inversly proportional to temperature T. i.e., when T increase, δ decrease and vice-versa.
Hence, δ-T grapgh will be a rectangular hyperbola as shown in the above figure.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The initial volume and pressure of a gas are V and P respectively. It is expanded through two different processes such t...A monoatomic gas at a pressure P, having a volume V expands isothermally to a volume 2 V and then adiabatically to a vol...Three rods made of the same material and having the same cross-section been joined as shown in the figure. Each rod is o...Two identical containers A and B with friction pistons contain the ideal gas at the same temperature and the same volume...The volume of an ideal gas increases 8 times and temperature becomes $(1/4)^{\text{th~}}$ of initial temperature during ...