ThermodynamicsHard
Question
A gas mixture consists of 2 moles of oxygen and 4 moles of argon at temperature T. Neglecting all vibrational modes, the total internal energy of the system is
Options
A.4 RT
B.15 RT
C.9 RT
D.11 RT
Solution
Internal energy of n moles of an ideal gas at temperature T is given by
U = n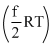
Where, f = degree of freedom
= 5 for O2 and 3 for Ar
Hence, U = UQ2 + UAr = 2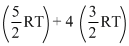 = 11RT
= 11RT
U = n
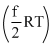
Where, f = degree of freedom
= 5 for O2 and 3 for Ar
Hence, U = UQ2 + UAr = 2
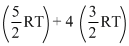 = 11RT
= 11RT Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In a given process of an ideal gas, dW = 0 and dQ...The wavelength of maximum energy released during an atomic explosin was 2.93 × 10-10m. The maximum temperature atta...The average translational kinetic energy of O2 (molar mass 32) molecules at a particular temperature is 0.048 eV. The tr...three rods of identical cross-sectional area and made from the same material from the sides of an isoscales traangles AB...If one mole of a monoatomic gas (γ = 5 / 3 ) is mixed with one mole of a diatomic gas (γ = 7 / 5 ), the value ...