ThermodynamicsHard
Question
The average translational kinetic energy of O2 (molar mass 32) molecules at a particular temperature is 0.048 eV. The translational kinetic energy of N2 (molar mass 28) molecules in eV at the same temperature is
Options
A.0.0015
B.0.003
C.0.048
D.0.768
Solution
Average translational kinetic energy of an ideal gas molecules is 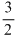 kT which depends ontemperature only.
kT which depends ontemperature only.
Therefore, if temperature is same, translational kinetic energy of O2 and N2 both will be equal.
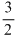 kT which depends ontemperature only.
kT which depends ontemperature only. Therefore, if temperature is same, translational kinetic energy of O2 and N2 both will be equal.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Helium gas goes through a cycle ABCDA(consisting of two isochoric and two isobaric lines) as shown in figure. Efficiency...For an ideal gas...A quantity of heat requirad to change the unit mass of a solid substance to its liquid state, while the temperature rema...In the above question, the curve for which the heat is absorbed from the surroundings is :-...One mole of an ideal diatomic gas expands from volume V to 2 V isothermally at a temperature $27^{\circ}C$ and does W jo...