ThermodynamicsHard
Question
If one mole of a monoatomic gas (γ = 5 / 3 ) is mixed with one mole of a diatomic gas (γ = 7 / 5 ), the value of γ for the mixture is
Options
A.1.40
B.1.50
C.1.53
D.3.07
Solution
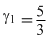 means gas is monoatomic or
means gas is monoatomic or 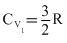
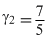 means gas is diatomic or
means gas is diatomic or 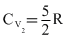
CV (of the mixture)
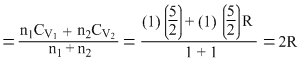
CP (of the mixture) = CV + R = 3R
∴ γmixture =
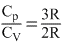 = 1.5
= 1.5∴ Correct option is (b).
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A gas mixture contain 1 gm. H2 and 1 gm. He if temp. of gas mixture increase 0oC to 100o C at isobaric process. Than fin...Amount of work which can be obtained from 200 cal heat, will be :...A spherical black body with a radius of 12 cm radiates 450 W power at 500 K. If the radius were halved and temperature d...In the diagrams (i) to (iv) of variation of volume with changing pressure is shown. A gas is taken along the path ABCDA....The figure shows the P-V plot of an ideal gas taken through a cycle ABCDA. The part ABC is a semi-circle and CDA is half...