ThermodynamicsHard
Question
Tw identical containers A and B with frictionless pistons contains the same ideal gas at the same temperature and the same volume V. The gas in each cylinder is now allowed to expand isothermally to the same final volume 2V. The changes in the pressure in A and B are found to be ᐃp and 1.5 ᐃp respectively. Then
Options
A.4mA = 9mB
B.2mA = 3mB
C.3mA = 2mB
D.9mA = 4mB
Solution
Process is isothermal. Therefore, T = constant. 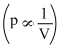 volume is increasing,therefore, pressure will decrease.
volume is increasing,therefore, pressure will decrease.
In chamber A →
- ᐃP = (PA)i - (PA)f =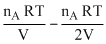
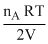 .....(i)
.....(i)
In chamber B →
-1.5ᐃp = (PB)i - (PB)f =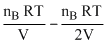 .....(ii)
.....(ii)
From eqs (i) and (ii)
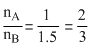
or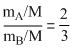 or
or 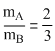
or 3mA = 2mB
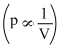 volume is increasing,therefore, pressure will decrease.
volume is increasing,therefore, pressure will decrease. In chamber A →
- ᐃP = (PA)i - (PA)f =
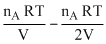
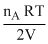 .....(i)
.....(i)In chamber B →
-1.5ᐃp = (PB)i - (PB)f =
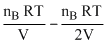 .....(ii)
.....(ii)From eqs (i) and (ii)
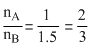
or
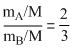 or
or 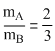
or 3mA = 2mB
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The volume of a gas is reced adiabatically to (1/4) of its volume at 27oC. If γ = 1.4.The new temrereacture will be...When a solid is converted into a gas, directly by heating then this process is known as :...As shown in the diagram for a closed path ABCA :...One mole of monoatomic gas is taken round the cyclic process. Heat absorbed by gas in process AB is :-...The thermal conductivity of a rod 2.What is its thermal resistivity ?...