ThermodynamicsHard
Question
The volume of a gas is reced adiabatically to (1/4) of its volume at 27oC. If γ = 1.4.The new temrereacture will be :
Options
A.300 × (4)0.4 K
B.150 × (4)0.4 K
C.250 × (4)0.4 K
D.none of these
Solution
For adiabatic change, The equation is TVγ-1 = constant
T1V1γ-1 = T2V2γ-1
(27 + 273) V2γ-1 = T2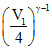
300 × V1γ-1 =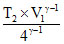 ⇒ T2 = 300 × 4γ-1
⇒ T2 = 300 × 4γ-1
T2 = 300 × 41.4-1 = 300 × 40.4 K
T1V1γ-1 = T2V2γ-1
(27 + 273) V2γ-1 = T2
300 × V1γ-1 =
T2 = 300 × 41.4-1 = 300 × 40.4 K
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
During the melting of a slab of ice at 273 K at atomospheric pressure...The relation Cp − Cv = R/J implies :-(J−Joule constant)...5.6 liter of helium gas at STP is adiabatically compressed to 0.7 lit. Taking the initial temperaure to be T1, the work ...Temperature of an ideal gas is 300 K. The change in temperature of the gas when its volume changes from V to 2V in the p...A system is brought from state A to B in an isometric change. The amount of heat absorbed by the system will be :-...