ThermodynamicsHard
Question
When you make ice cubes, the entropy of water
Options
A.dose not change
B.increases
C.decreases
D.may either increases depending on the process if used
Solution
dS = 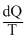
In freezing process dQ is neative so entropy decreass.
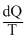
In freezing process dQ is neative so entropy decreass.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
If 2 gm moles of a diatomic gas and 1 gm mole of a mono-atomic gas are mixed then the value of (γ = Cp/Cv) for mixt...A system is brought from state A to B in an isometric change. The amount of heat absorbed by the system will be :-...A sample of gas expands from volume V1 to V2 The amount of work done by the gas is greast, when the expansion is :...The volume of a 2 mole ideal gas at 27oC is V. It expands adiabatically to the volume 2V. If γ = 5/3, R = 8.31 J/mo...Which of the following is incorrect regarding the first law of thermodynamics?...