Mole ConceptHard
Question
10 L of hard water required 0.56 g of lime (CaO) for removing hardness. Hence, temporary hardness in ppm (part per million 106) of CaCO3 is:
Options
A.100
B.200
C.10
D.20
Solution
Temporary hardness is due to HCO3- of Ca2+ and Mg2+
Ca(HCO3)2 +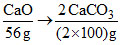 + H2O
+ H2O
mole of CaCO3 = 2
Ca(HCO3)2 +
mole of CaCO3 = 2
ppm of CaCO3 = (2/104)×106 =200
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Which of the following(s) is/are correct statement?...Law of multiple proportions is not applicable for the oxide(s) of...A nvesel conains 14 g (moles) of hydrogen and 96 g (3 moles) of oxygen at ATP. Chemical reaction induced by passing elec...An amount of 5 moles of A, 6 moles of B and excess amount of C are mixed to produce a final product D, according to the ...If 240 g of carbon is taken in a container to convert it completely to CO2 but in industry it has been found that 280 g ...