SolutionHard
Question
The amount of electricity that should be passed through CuSO4 solution with Cu electrodes to deposit 0.1 gram atom of Cu
Options
A.9650 Coulombs
B.96500 Coulombs
C.19300 Coulombs
D.193000 Coulombs
Solution
Weight of Cu = mole × atomic weight
= 0.1 × 63.5 = 6.35 g
According to Faraday law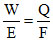
∴ Q =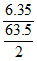 × 96500 cb
× 96500 cb
= 19300 cb
= 0.1 × 63.5 = 6.35 g
According to Faraday law
∴ Q =
= 19300 cb
Create a free account to view solution
View Solution FreeMore Solution Questions
The boiling point of an azeotropic mixture of water and ethanol is less than that of water and ethanol, separately. The ...A solution of sulphuric acid in water exhibits :...80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual ga...A solution of ‘x’ mole of sucrose in 100 g of water freezes at − 0.2°C. As ice separates out, the freezing point goes do...Which of the following is less than zero for ideal solutions ?...