KTGHard
Question
The molecules of mass m, moving with velocity v are absorbed after colliding with walls of a container. If the number of molecules per unit volume of the gas is n, then the pressure exerted by the gas will be-
Options
A.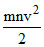
B.2mnv2
C.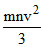
D.mnv2
More KTG Questions
If the total number of H2 molecules is double that of the O2 molecules then the ratio of total kinetic energies of H2 to...The temperature at which the r.m.s velocity of oxygen molecules equal that of nitrogen molecules at 100oC is nearly:...One mole of an ideal gas at a temperature T1K expands slowly according to the law = constant. Its final temperature is T...Critical temperature is that temperature...Let P1 and P2 be the final pressure of the samples 1 and 2 respectively in the previous question then :...