KTGHard
Question
The specific heats of argon at constant pressure and constant volume are 525 J/kg and 315 J/kg respectively. Its density at NTP will be :-
Options
A.1.77 kg/m3
B.0.77 kg/m3
C.1.77 gm/m3
D.0.77 gm/m3
Solution
Cp - Cv = 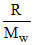
525 - 315 = 210 =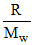
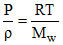
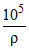 = 210 × 273
= 210 × 273
ρ = 1.77 kg/m3
525 - 315 = 210 =
ρ = 1.77 kg/m3
Create a free account to view solution
View Solution FreeMore KTG Questions
Graph shows a hypothetical speed distribution for a sample of N gas particle (for V > V0; = 0)...A real gas behaves like an ideal gas if its...The pressure exerted by a gas in P0. If the mass of molecules becomes half and their velocities become double the pressu...A cylinder of 200 litre capacity is containing H2. The total average translational kinetic energy of molecules is 1.52 &...Four containers are filled with mono atomic ideal gases. For each container, the number of moles, the mass of an individ...