Analytical ChemistryHard
Question
27 g of aluminium react completely with how many grams of oxygen ?
Options
A.8g
B.16g
C.32g
D.24g
Solution
2Al + 3/2O2 → Al2O3
i.e., 2 × 27g Al =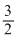 × 32gO2
× 32gO2
or 27g Al =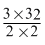 = 24g O2
= 24g O2
i.e., 2 × 27g Al =
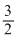 × 32gO2
× 32gO2or 27g Al =
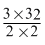 = 24g O2
= 24g O2 Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Upon treatment with ammonical H2S , the metal ion that precipitates as a sulfide is -...A white crystalline solid (A) on boiling with caustic soda solution gave a gas (B), which when passed through an alkalin...Which of the following is insoluble in dil. HNO3 but dissolves in aquaregia ?...Cu2+ ions give white precipitate with :...The composition of golden spangles is :...