Mole ConceptHard
Question
A gaseous mixture contains oxygen and nitrogen in the ratio of 1 : 4 by weight. Therefore, the ratio of their number of molecules is :
Options
A.1 : 4
B.1 : 8
C.7 : 32
D.3 : 16
Solution
Number of molecules (N) ∞ number of moles (n)
or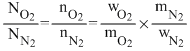
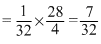
or
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Elements P and Q form two types of non-volatile, non-ionizable compounds PQ and ${PQ}_{2}$. When 1 g of PQ is dissolved ...An amount of 1.0 × 10–3 moles of Ag+ and 1.0 × 10–3 moles of CrO4 2− reacts together to form solid Ag2CrO4. What is the ...Two elements A and B combine to form compound X and Y. For the fixed mass of A, masses of B combined for the compounds X...Depression of freezing point of 0.01 molal aq. CH3COOH solution is 0.02046. 1 molal urea solution freezes at -1.86oC. As...Which expansion will produce more change in entropy during reversible & isothermal process?...