SolutionHard
Question
On mixing 3g of non-volatile solute in 200 mL of water, its Boiling point (100oC) be comes 100.52oC. If Kb for water is 0.6 K/n then molecular wt. of solute is :
Options
A.10.5 mol-1
B.12.6 mol-1
C.15.7 mol-1
D.17.3 mol-1
Solution
ᐃT = Kb 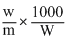
0.52 = 0.6 ×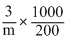
m =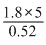 = 17.3g mole-1
= 17.3g mole-1
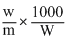
0.52 = 0.6 ×
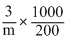
m =
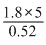 = 17.3g mole-1
= 17.3g mole-1Create a free account to view solution
View Solution FreeMore Solution Questions
Which one of the following statements is correct when SO2 is passed through acidified K2Cr2O7 solution?...An ionic compound that attracts atmospheric water so strongly that a hydrate is formed is said to be :...For a dilute solution containing 2.5 g of a non-volatile non-electrolyte solute in 100 g of water, the elevation in boil...Among the following the surfactant that will form micelles in aqueous solution at the lowest molar concentration at ambi...A binary liquid solution is prepared by mixing n-heptane and ethanol. Which one of the following statements is correct r...