Chemical Kinetics and Nuclear ChemistryHard
Question
Consider the chemical reaction,
N2(g) + 2H2(g) → 2NH3(g)
The rate of this reaction can be expressed in terms of time derivatives of concentration of
N2(g), H2(g), or NH3(g). Identify the correct relationship amongst the rate expressions :
N2(g) + 2H2(g) → 2NH3(g)
The rate of this reaction can be expressed in terms of time derivatives of concentration of
N2(g), H2(g), or NH3(g). Identify the correct relationship amongst the rate expressions :
Options
A.Rate = 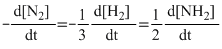
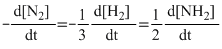
B.Rate = 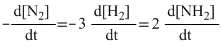
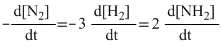
C.Rate = 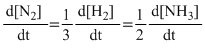
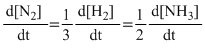
D.Rate = 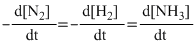
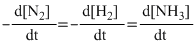
Solution
For a general reaction, aA + bB → cC + dD
Rate =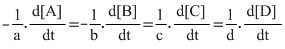
N2 + 3H2 → 2NH3
Rate =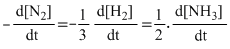
Rate =
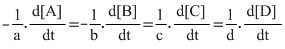
N2 + 3H2 → 2NH3
Rate =
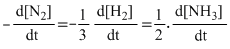
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Sometimes, reaction rates can be estimated by knowing...The activation energy of a reaction can be lowered by...For the reaction A (g) + 2B(g) → C(g) + D(g) = k [A] [B]2 Initial pressure of A and B are respectively 0.60 atm an...The energies of activation for forward and reverse reactions for A2 + B2 ⇋ 2AB are 180 kJ mol-1 2AB are 180 kJ mol...Which of the following statement(s) is/are correct for a photochemical reaction?...