Solid StateHard
Question
How many unit cell are present in a cube-shaped ideal crystal of NaCl of mass 1.00 gm. [Atomic masses : Na = 23, Cl = 35.5]
Options
A.2.57 × 1021 unit cells
B.5.14 × 1021 unit cells
C.1.25 × 1021 unit cells
D.1.71 × 1021 unit cells
Solution
No. of moles in 1g = 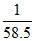
So. number of molecules =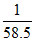 × NA
× NA
∵ 4 molecules are present in one unit cell of NaCl.
∴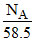 molecule, are present in =
molecule, are present in = 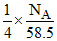
= 2.57 × 1021 unit cell of N
So. number of molecules =
∵ 4 molecules are present in one unit cell of NaCl.
∴
= 2.57 × 1021 unit cell of N
Create a free account to view solution
View Solution FreeMore Solid State Questions
Antifluorite structure is derived from fluorite structure by :...Which of the following is/are correct regarding point defects in solids?...Sapphire is aluminum oxide. Aluminum oxide crystallizes with aluminum ions in two-thirds of the octahedral voids in the ...Platinum crystallises in a face centered cube crystal with a unit cell length of 3.9231 A0. The density and atomic radiu...In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the for...