Solid StateHard
Question
In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the formula of the compound would be if two atoms of X are missing from the corners :
Options
A.X4Y
B.X3Y4
C.XY4
D.X2Y4
Solution
for X, 6 × 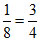
for Y, 6 ×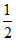 = 3
= 3
So X3/4 Y3 or X3 Y12 or XY4
for Y, 6 ×
So X3/4 Y3 or X3 Y12 or XY4
Create a free account to view solution
View Solution FreeMore Solid State Questions
Packing fraction in 2D-hexagonal arrangement of identical spheres is...A metal exists as FCC crystal. If the atomic radius is 100$\sqrt{2}$ pm and the density of metal is 12,500 kg/m3, then t...A transition metal M can exist in two oxidation states +2 and +3. It forms an oxide whose experimental formula is given ...The number of octahedral void(s) per atom present in a cubic close-packed structure is...The composition of a sample of cuprous oxide is found to be $\text{Cu}_{1.92}\text{O}_{1.00}$ due to a metal deficiency ...