Ionic EquilibriumHard
Question
Which of the following acids has the smallest dissociation constant ?
Options
A.CH3CHFCOOH
B.FCH2CH2COOH
C.BrCH2CH2COOH
D.CH3CHBrCOOH
Solution
Stronger is the acid higher is its dissociation constant for the given acids, the order of acidic strength is
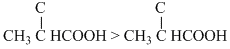 > FCH2CH2COOH > BrCH2CH2COOH
> FCH2CH2COOH > BrCH2CH2COOH
Thus smallest dissociation constant will be for BrCH2CH2COOH
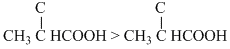 > FCH2CH2COOH > BrCH2CH2COOH
> FCH2CH2COOH > BrCH2CH2COOHThus smallest dissociation constant will be for BrCH2CH2COOH
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which one is the correct graph (figure) for the corresponding acid base titration?...What will be the effect of adding 100 ml of 0.001 M-HCl solution to 100 ml of a solution having 0.1 M-HA? The acid disso...The pH of water is 7.0 at 25oC. If water is heated to 70oC, then...How many grams of NaOH should be added in 500 ml of 2 M acetic acid solution to get a buffer solution of maximum buffer ...The phenomenon of optical activity will be shown by :-...