Ionic EquilibriumHard
Question
The precipitate of CaF2 (Ksp = 1.7 × 10-10) is obtained, when equal volumes of the following are mixed :
Options
A.10-10 M Ca2+ + 10-4MF-
B.10-2 M Ca2+ + 10-3MF-
C.10-5 M Ca2+ + 10-3MF-
D.10-3 M Ca2+ + 10-5MF-
Solution
Precipitation will be started when Ionic product > solubility product i, e, when
[Ca2+][F-]2 > Ksp
When equal volumes of 10-2 MCa2+ and 10-3MF- solutions are mixed then
Ionic product =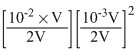
= 1.25 × 10-9
It exceeds Ksp (1.7 × 10-9), thus precipitation will be started.
For all other cases, Ionic product < Ksp
[Ca2+][F-]2 > Ksp
When equal volumes of 10-2 MCa2+ and 10-3MF- solutions are mixed then
Ionic product =
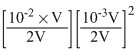
= 1.25 × 10-9
It exceeds Ksp (1.7 × 10-9), thus precipitation will be started.
For all other cases, Ionic product < Ksp
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
What concentration of free CN– must be maintained in a solution that is 1.8 M-AgNO3 and 0.16 M-NaCl to prevent AgCl from...The pKa of acetylasliclic acid (aspirin) is 3.5. The pH of gastric juice in human stomach is about 2-3 and the pH in the...One litre of solution contains 10-5 moles of H+ ions at 25oC. Percentage ionisation of water in solution is :...The solubility product of AgC2O4 at 25°C is 2.3 × 10–11 M3. A solution of K2C2O4 containing 0.15 moles in 500 ml water i...Calcium lactate is a salt of weak acid and represented as Ca(Lac)2. A saturated solution of Ca(Lac)2 contains 0.125 mole...