Ionic EquilibriumHard
Question
One litre of solution contains 10-5 moles of H+ ions at 25oC. Percentage ionisation of water in solution is :
Options
A.1.8 × 10-7 %
B.1.8 × 10-9 %
C.3.6 × 10-9 %
D.1.8 × 10-11 %
Solution
In this solution, source of OH- is water
∴ Cα = [OH-]
α =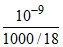 = 1.8 × 10-11 M
= 1.8 × 10-11 M
% ionisation = 1.8 × 10-9 M
∴ Cα = [OH-]
α =
% ionisation = 1.8 × 10-9 M
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Aqueous solutions of HNO3, KOH, CH3COOH, and CH3COONa of identical concentrations are provided. The pair (s) of solution...Which of the following relation(s) is/are true for alkaline solution?...The amino acid glycine (NH2CH2COOH) is basic because of its –NH2 group and acidic because of its –COOH group. By a proce...The solubility product of Mg(OH)2 is 9.0 × 10−12. The pH of an aqueous saturated solution of Mg(OH)2 is (log 1.8 = 0.26,...A certain buffer solution contains equal concentration of X- and HX. The Kb for X- is 10-10. The pH of the buffer is :...