Mole ConceptHard
Question
Equal volume of 0.1M NaCl and 0.1M FeCl2 are mixed with no change in volume due to mixing. Which of the following will be true for the final solution. (No precipitation occurs). Assume complete dissociation of salts and neglect any hydrolysis.
Options
A.[Na+] = 0.05 M
B.[Fe2+] = 0.05M
C.[Cl-] = 0.3M
D.[Cl-] = 0.15M
Solution
Vml 0.1 M NaCl
Vml 0.1 M FeCl2
[Na+] =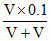 = 0.05 M
= 0.05 M
[Fe2+] =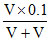 = 0.05 M
= 0.05 M
[Cl-] =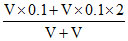 = 0.15 M
= 0.15 M
Vml 0.1 M FeCl2
[Na+] =
[Fe2+] =
[Cl-] =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
An aqueous solution containing KIO3 was treated with an excess of KI solution. The solution is acidified with HCl. The l...Cl - CH2 - CH2 - CH2 - Br + KCN → ? (1 mole) (1 mole)Product will be :-...Given the polymers (i) Nylon 6,6 (ii) Buna-S, (iii) Polythene, arrange these in increasing order of their inter-molecula...The density of air is 0.001293 g/cm3 at STP. Identify which of the following statement is correct...Calculate the mass percent (w/w) of sulphuric acid in a solution prepared by dissolving 4 g of sulphur trioxide in a 100...