Mole ConceptHard
Question
100 mL of 0.06 M Ca(NO3)2 is added to 50 mL of 0.06 M Na2C2O4. After the reaction is complete.
Options
A.0.003 moles of calcium oxalate will get precipitated.
B.0.003 M of excess Ca2+ will remain in excess.
C.Na2C2O4 is the limiting reagent.
D.Ca(NO3)2 is the excess reagent.
Solution
Ca(NO3)2 + Na2C2O4 → CaC2O4 ↓ + 2NaNO3
100 × 0.06 50 × 0.06
= 6 m mol = 3 m mol 3 m mol = 0.003 mol
Na2C2O4 is the limiting reagent.
∴ 3 m mol Na2C2O4 ≡ 3 m mol Ca(NO3)2
≡3 m mol CaC2O4 ≡ 6 m mol NaNO3
m mol of Ca(NO3)2 left = 6 - 3 = 3 m mol = 0.003 mol
MCa2+(left) =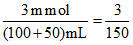 = 0.02M
= 0.02M
Hence, option (b) is wrong.
100 × 0.06 50 × 0.06
= 6 m mol = 3 m mol 3 m mol = 0.003 mol
Na2C2O4 is the limiting reagent.
∴ 3 m mol Na2C2O4 ≡ 3 m mol Ca(NO3)2
≡3 m mol CaC2O4 ≡ 6 m mol NaNO3
m mol of Ca(NO3)2 left = 6 - 3 = 3 m mol = 0.003 mol
MCa2+(left) =
Hence, option (b) is wrong.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Cl - CH2 - CH2 - CH2 - Br + KCN → ? (1 mole) (1 mole)Product will be :-...In which mode of expression, the concentration of a solution remains independent of temperature ?...Fraction of molecules at a given temperature for a gas is in the order at -...1.2575 g sample of [Cr(NH3)6]SO4Cl(Mw = 251.5) is dissolved to prepare 250mL solution showing and osmotic pressure of 1....100 gm impure CaCO3 on heating gives 5.6 lt. CO2 gas at STP. Find the percentage of calcium in the lime stone sample. [A...