Mole ConceptHard
Question
Minimum amount of Ag2CO3 (s) required to produce sufficient oxygen for the complete combustion of C2H2 which produces 11.2 ltr of CO2 at S.T.P after combustion is: [Ag = 108]
Ag2CO3 (s) → 2Ag (s) + CO2 (g) + O2 (g)
C2H2 + 5/2O2 → 2CO2 + H2O
Ag2CO3 (s) → 2Ag (s) + CO2 (g) + O2 (g)
C2H2 + 5/2O2 → 2CO2 + H2O
Options
A.276 g
B.345 g
C.690 g
D.1380 g
Solution
Ag2CO3 (s) → 2Ag (s) + CO2 (g) + 1/2O2 (g)
C2H2 + 5/2O2 → 2CO2 + H2O
By Stoichiometry of reaction
Moles of CO2 formed =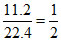
Moles of O2 required =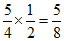
∴ Moles of Ag2CO3 required = 2 ×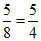
Mass of Ag2CO3 required =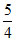 × 276 = 345 g
× 276 = 345 g
C2H2 + 5/2O2 → 2CO2 + H2O
By Stoichiometry of reaction
Moles of CO2 formed =
Moles of O2 required =
∴ Moles of Ag2CO3 required = 2 ×
Mass of Ag2CO3 required =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
100 mL of 0.06 M Ca(NO3)2 is added to 50 mL of 0.06 M Na2C2O4. After the reaction is complete....Dopamine is a neurotransmitter, a molecule that serves to transmit message in the brain. The chemical formula of dopamin...Match the coloum :...A compound having the empirical formula, C3H4O has a molecular weight of 170 ± 5. The molecular formula of the compound ...Which of the following is/are examples of uni molecular reactions ?...