Mole ConceptHard
Question
64 g of an organic compound has 24 g carbon and 8 g hydrogen and the rest is oxygen. The empirical formula of the compound is
Options
A.CH4O
B.CH2O
C.C2H4O
D.None
Solution
C H O
mass 24 8 32
moles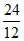
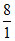
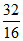
ratio 2 8 2
Simple integer ratio 1 4 1
Hence empirical formula is CH4O
mass 24 8 32
moles
ratio 2 8 2
Simple integer ratio 1 4 1
Hence empirical formula is CH4O
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
∠FAsF bond angle in AsF3Cl2 molecule is :-...If we consider that , in place of ; mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mo...A solution containing 0.1 mol of a metal chloride MClx requires 500 ml of 0.8 M AgNO3 solution for complete reaction MCl...Which of the following statements is/are correct ?...The volume strenght of 1.5 N H2O2 solution is :...