Mole ConceptHard
Question
A solution containing 0.1 mol of a metal chloride MClx requires 500 ml of 0.8 M AgNO3 solution for complete reaction MClx + xAgNO3 → xAgCl + M(NO3)x. Then the value of x is
Options
A.1
B.2
C.4
D.3
Solution
MClx + x AgNO3 → xAgCl + M (NO3)x
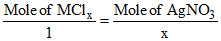
0.1 =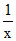 (0.5 × 0.8)
(0.5 × 0.8)
x =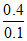 = 4
= 4
0.1 =
x =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The pair of species having identical shapes for molecules of both species is...Fraction of molecules at a given temperature for a gas is in the order at -...Total charge required for the oxidation of two moles Mn3O4 into MnO4-2 in presence of alkaline medium is :-...1.2575 g sample of [Cr(NH3)6]SO4Cl(Mw = 251.5) is dissolved to prepare 250mL solution showing and osmotic pressure of 1....1 volume of gaseous compound consisting C, H, O on complete combustion in presence of 2.5 volume of O2 gives 2 vol. of s...