Mole ConceptHard
Question
Which of the following statements is/are correct ?
Options
A.21.2 g sample of impure Na2CO3 is dissolved and reacted with a solution of CaCl2, the weight of precipitate of CaCO3 is 10.0 g. the % purity of Na2CO3 is 50%.
B.The percentage purity of Na2CO3 is 60%
C.The number of moles of Na2CO3 = CaCO3 = 0.1 mol.
D.The number of moles of NaCl formed is 0.1 mol.
Solution
Mw of CaCO3 = 100, Mw of Na2CO3 = 106
Mw of HNO3 = 63 g mol-1
Na2CO3 + CaCl2 → CaCO3 + 2NaCl
a. moles of CaCO3 =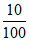 = 0.1 mol
= 0.1 mol
moles of Na2CO3 = moles of CaCO3 ≡ 2 × moles of NaCl
Weight of Na2CO3 = 0.1 × 106 = 10.6 g
% purity Na2CO3 =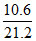 × 100 = 50%
× 100 = 50%
b. Wrong.
c. Correct.
d. moles of NaCl = 2 × 0.1 = 0.2 mol
Mw of HNO3 = 63 g mol-1
Na2CO3 + CaCl2 → CaCO3 + 2NaCl
a. moles of CaCO3 =
moles of Na2CO3 = moles of CaCO3 ≡ 2 × moles of NaCl
Weight of Na2CO3 = 0.1 × 106 = 10.6 g
% purity Na2CO3 =
b. Wrong.
c. Correct.
d. moles of NaCl = 2 × 0.1 = 0.2 mol
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
V1 ml of unknown gas (A) + V2 ml of O2 → (V1 + V2) ml of CO2. Gas ‘A’ may be...A person needs an average of 2.0 mg of riboflavin (vitamin B2) per day. How many grams of butter should be taken by the ...Dissolving 120g of urea (molecular wt. 60) in 1000g of water gave a solution of density 1.15 g/ml. The molarity of the s...Large quantities of ammonia are burned in the presence of a platinum catalyst to give nitric oxide, as the first step in...Two successive reactions, A → B and B → C, have yields of 90% and 80%, respectively. What is the overall percentage yiel...