Mole ConceptHard
Question
5.85 g of NaCl is dissolved in 1 L of pure water. The number of ions in 1 mL of this solution is
Options
A.6.02 × 1019
B.1.2 × 1022
C.1.2 × 1020
D.6.02 × 1020
Solution
Mole of NaCl = 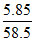 = 0.1
= 0.1
Molarity =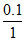 = 0.1 M
= 0.1 M
Moles in 1 ml of solution = MV = 0.1 × 10-3 = 10-4 mole.
Number of ions in 1 ml = 2 × 10-4 × 6.023 × 1023 = 1.204 × 1020.
Molarity =
Moles in 1 ml of solution = MV = 0.1 × 10-3 = 10-4 mole.
Number of ions in 1 ml = 2 × 10-4 × 6.023 × 1023 = 1.204 × 1020.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
CH3-CO-CH3(g) ⇋ CH3-CH3 (g) + CO(g)Initial pressure of CH3COCH3 is 100 mm. When equilibrium is set up mole fractio...The process by which organisms with different evolutionary history evolve similar phenotypic adaptation in response to a...Two solutions of a substance (non electrolyte) are mixed in the following manner. 480 ml of 1.5 M first solution + 520 m...Common salt obtained from sea - water contains 96% NaCl by mass. The approximate number of molecules of NaCl present in ...The total number of electrons in one molecule of carbon dioxide is :...