Mole ConceptHard
Question
Common salt obtained from sea - water contains 96% NaCl by mass. The approximate number of molecules of NaCl present in 10.0 g of the common salt is : (At. wt. Na = 23)
Options
A.1021
B.1022
C.1023
D.1024
Solution
Mass of NaCl = 10 × 0.96 = 9.6 gm
moles of NaCl =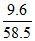
no. of molecules =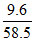 × 6.023 × 1023 ≃ 1023
× 6.023 × 1023 ≃ 1023
moles of NaCl =
no. of molecules =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
A quantity of 5 g of a crystalline salt when rendered anhydrous lost 1.8 g of water. The formula mass of the anhydrous s...The energy of activation is :-...In a textile mill, a double effect evaporator system concentrates weak liquor containing 4% (by mass) caustic soda to pr...The strength of 10-2 M Na2CO3 solution in terms of molality will be (density of solution = 1.10 g mL-1). (Molecular weig...Two solutions of a substance (non electrolyte) are mixed in the following manner. 480 ml of 1.5 M first solution + 520 m...