Mole ConceptHard
Question
A 10.0 g sample of a mixture of calcium chloride and sodium chloride is treated with Na2CO3 solution. This calcium carbonate is heated to convert all the calcium to calcium oxide and the final mass of calcium oxide is 1.62 gm. The percentage by mass of calcium chloride in the original mixture is -
Options
A.15.2%
B.32.1%
C.21.8%
D.11.07%
Solution
CaCl2 + Na2CO3 → CaCO3 + 2 NaCl
CaCO3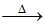 CaO + CO2
CaO + CO2
Mole of CaCl2 = mole of CaCO3 = mole of CaO =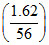
Mass of CaCl2 =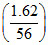 Molar mass of CaCl2
Molar mass of CaCl2
=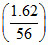 × 111 gm.
× 111 gm.
% of CaCl2 =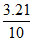 × 100 = 32.1 %.
× 100 = 32.1 %.
CaCO3
Mole of CaCl2 = mole of CaCO3 = mole of CaO =
Mass of CaCl2 =
=
% of CaCl2 =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
100 mL of PH3 on decomposition produced phosphorous and hydrogen. The change in volume is :-...In the reaction2Al(s) + 6HCl(aq) → 2Al3+(aq) + 6Cl-(aq) + 3H2(g) :-...If 240 g of carbon is taken in a container to convert it completely to CO2 but in industry it has been found that 280 g ...In a binary ideal liquid solution of A and B in which vapour pressure of pure component A and B are 600 and 400 torr res...The molar concentration of HCl (aq.) is 10-5 M. Which of the following statements are correct.(dsolution = 1 gm /cc)...