Mole ConceptHard
Question
How many moles of magnesium phosphate, Mg3(PO4)2 will contain 0.25 mole of oxygen atoms?
Options
A.0.02
B.3.125 × 10-2
C.1.25 × 10-2
D.2.5 × 10-2
Solution
Mg3(PO4)2
′n′ moles
8n = 0.25
n =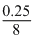
=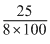 = 3.125 × 10-2
= 3.125 × 10-2
′n′ moles
8n = 0.25
n =
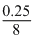
=
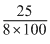 = 3.125 × 10-2
= 3.125 × 10-2Create a free account to view solution
View Solution FreeMore Mole Concept Questions
If the yield of chloroform obtainable from acetone and bleaching powder is 75%, then what mass of acetone is required fo...When burnt in air, 14.0 g mixture of carbon and sulphur gives a mixture of CO2 and SO2 in the volume ratio of 2: 1, the ...Which of the following is dependent on temperature?...In Carius method, 0.75 g of an organic compound gave 1.2 g of barium sulphate, find percentage of sulphur (molar mass )....Law of multiple proportions is not applicable for the oxide(s) of...