Mole ConceptHard
Question
If 10 g of Ag reacts with 1 g of sulphur, the amount of Ag2S formed will be [Atomic weight of Ag = 108, S = 32] ?
Options
A.7.75 g
B.0.775 g
C.11 g
D.10 g
Solution
(A) Explanation : 2 Ag + S → Ag2 S
2 × 108 g of Ag reacts with 32 g of sulphur
10 g of Ag reacts with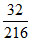 × 10 =
× 10 = 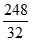 > 1 g
> 1 g
It means ′S′ is limiting reagent
32 g of S reacts to form 216 + 32 = 248 g of Ag2S
1 g of S reacts to form =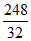 = 7.75 g
= 7.75 g
Alternately
neq of Ag =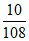 = 0.0925 neq of S =
= 0.0925 neq of S = 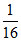 = 0.0625 (neq = number of equivalents)
= 0.0625 (neq = number of equivalents)
Since neq of S is less than neq of Ag
⇒ 0.0625 eq of Ag will react with 0.0625 eq of S to form 0.0625 eq of Ag2S
Hence, amount of Ag2S = neq × Eq. wt. of Ag2S
= 0.0626 × 124 = 7.75 g
2 × 108 g of Ag reacts with 32 g of sulphur
10 g of Ag reacts with
It means ′S′ is limiting reagent
32 g of S reacts to form 216 + 32 = 248 g of Ag2S
1 g of S reacts to form =
Alternately
neq of Ag =
Since neq of S is less than neq of Ag
⇒ 0.0625 eq of Ag will react with 0.0625 eq of S to form 0.0625 eq of Ag2S
Hence, amount of Ag2S = neq × Eq. wt. of Ag2S
= 0.0626 × 124 = 7.75 g
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The empirical formula of compound is CH2O. 0.084 moles of the compound contains 1.0 g of hydrogen. Moleculoar formula of...The hourly energy requirement of an astronaut can be satisfied by the energy released when 34 g of sucrose (C12H22O11) a...Which of the following has same mass...Calculate the N−N bond energy in N2H4 from given data.εN-H = 393 kJ/moleεH-H = 436 kJ/moleᐃHvap[N2...2SO2 + O2 ⇋ 2SO3 initially 4 moles each of SO2 & O2 are present, at equilibrium 25% of O2 is used. Total mols at e...