Mole ConceptHard
Question
The hourly energy requirement of an astronaut can be satisfied by the energy released when 34 g of sucrose (C12H22O11) are burnt in his body. How many g of oxygen would be needed to be carried in space capsule to meet his requirement for one day -
Options
A.916.2 gm
B.91.62 gm
C.8.162 gm
D.9.162 gm.
Solution
Use reaction C12H22O11 + 12O2 → 12CO2 + 11H2O.
In 24 hr. moles of sucrose consumed =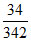 × 24.
× 24.
∴ In 24 hr. moles of O2 required =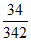 × 24 × 12. (according to stoichiometry).
× 24 × 12. (according to stoichiometry).
mass of O2 required =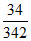 × 24 × 12 × 32 = 916.2 gm.
× 24 × 12 × 32 = 916.2 gm.
In 24 hr. moles of sucrose consumed =
∴ In 24 hr. moles of O2 required =
mass of O2 required =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
When a certain amount of octane, C8H18, is burnt completely, 7.04 g CO2 is formed. What is the mass of H2O formed simult...The number of g-molecules of oxygen in 6.023 × 1024 CO molecules is...The percentage by volume of C3H8 in a mixture of C3H8, CH4 and CO is 36.5. The volume of CO2 produced when 100 ml of the...The mass composition of universe may be given as 90% H2 and 10% He. The average molecular mass of universe should be...64 g of an organic compound has 24 g carbon and 8 g hydrogen and the rest is oxygen. The empirical formula of the compou...