JEE AdvancedChemical BondingHard
Question
Which of the following have identical bond order :
Options
A.CN-
B.O2-
C.NO+
D.CN+
Solution
Cn- (14), O2- (17), NO+ (14), CN+ (12)
Cn- : σls2, σ*ls2, σ2s2, σ*2s2,[ π2p2y = π2p2z] σ2p2x
Bond order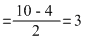
O2- : σls2, σ*ls2, σ2s2, σ*2s2, σ2p2x [ π2p2y = π2p2z] [π* 2p2y= π* 2p1z]
Bond order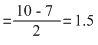
NO+ : σls2, σ*ls2, σ2s2, σ*2s2, σ2p2x [ π2p2y = π2p2z]
Bond order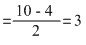
CN+ : σls2, σ*ls2, σ2s2, σ*2s2,[ π2p2y = π2p2z] σ2p2x
Bond order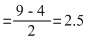
Cn- : σls2, σ*ls2, σ2s2, σ*2s2,[ π2p2y = π2p2z] σ2p2x
Bond order
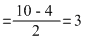
O2- : σls2, σ*ls2, σ2s2, σ*2s2, σ2p2x [ π2p2y = π2p2z] [π* 2p2y= π* 2p1z]
Bond order
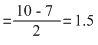
NO+ : σls2, σ*ls2, σ2s2, σ*2s2, σ2p2x [ π2p2y = π2p2z]
Bond order
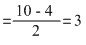
CN+ : σls2, σ*ls2, σ2s2, σ*2s2,[ π2p2y = π2p2z] σ2p2x
Bond order
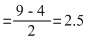
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The maximum radius of sphere that can be fitted in the octahedral hole of cubical closed packing of sphere of radius r i...The electrical conductivity of semiconductors:...According to molecular orbital theory, which of the following will not be a viable molecule ?...In a closed packed structure of mixed oxides, the lattice is composed of mixed oxides ions. One-eighth of tetrahedral vo...Which of the following comound is covalent ?...