JEE Main | 2018Chemical BondingHard
Question
According to molecular orbital theory, which of the following will not be a viable molecule ?
Options
A.
He2+
B.
H2-
C.
H22-
D.
He22+
Solution
The electronic configuration of is
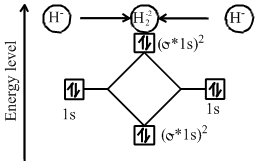
Bond order of
Hence does not exist, due to zero bond order.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
major product...Deduce from the following E - values of half cells, what combination of two half cell would results in a cell with the l...The major product formed in the following reaction is : →HeatHI...Which one of the following molecules contains no π bond ?...The decreasing values of bond angles from NH3 (106o) to SbH3 (101o) down group-15 of the periodic table is due to...